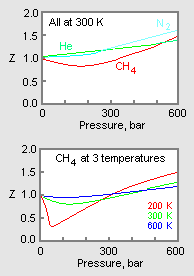
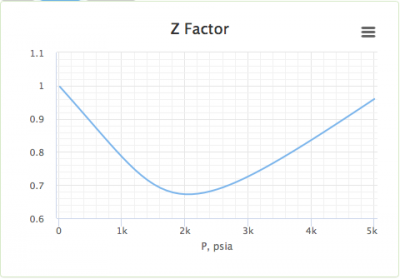
The graph of compressibility factor (Z) vs. P for one mole of a real gas ..
Solution For The graph of compressibility factor (Z) vs. P for one mole of a real gas is shown in following diagram. The graph is plotted at constant temperature 273K. If the slope of graph at very hi
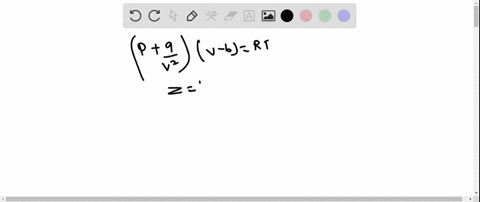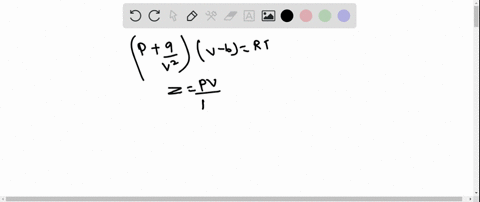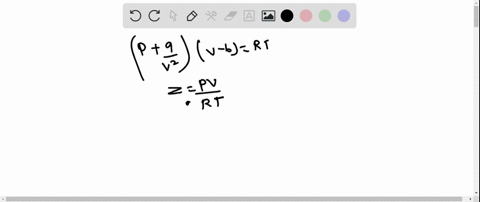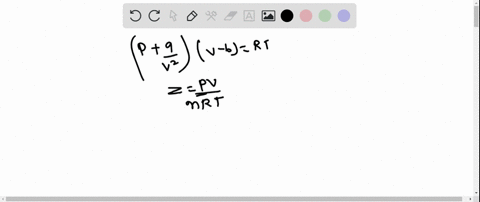
The graph of compressibility factor (Z) vs. P for one mole of a real gas is shown in following diagram. The graph is plotted at constant temperature 273K. If the slope of graph at very high pressure (dPdZ) is (2.81)atm−1 , then calculate volume of one mole of real gas molecules (in L/mol) Given : NA=6×1023 and R=27322.4LatmK−1mol−1

Compressibility Factor Calculator - File Exchange - MATLAB Central

The graph of compressibility factor (Z) vs. P for one mole of a
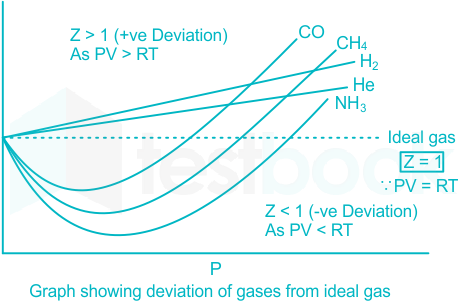
Solved] The compressibility factor for an ideal gas is

The graph of compressibility factor (Z) vs. P for one mole of a
In the following compressibility factor (Z) vs pressure graph at

Real gas z-Factor chart [2] Download Scientific Diagram
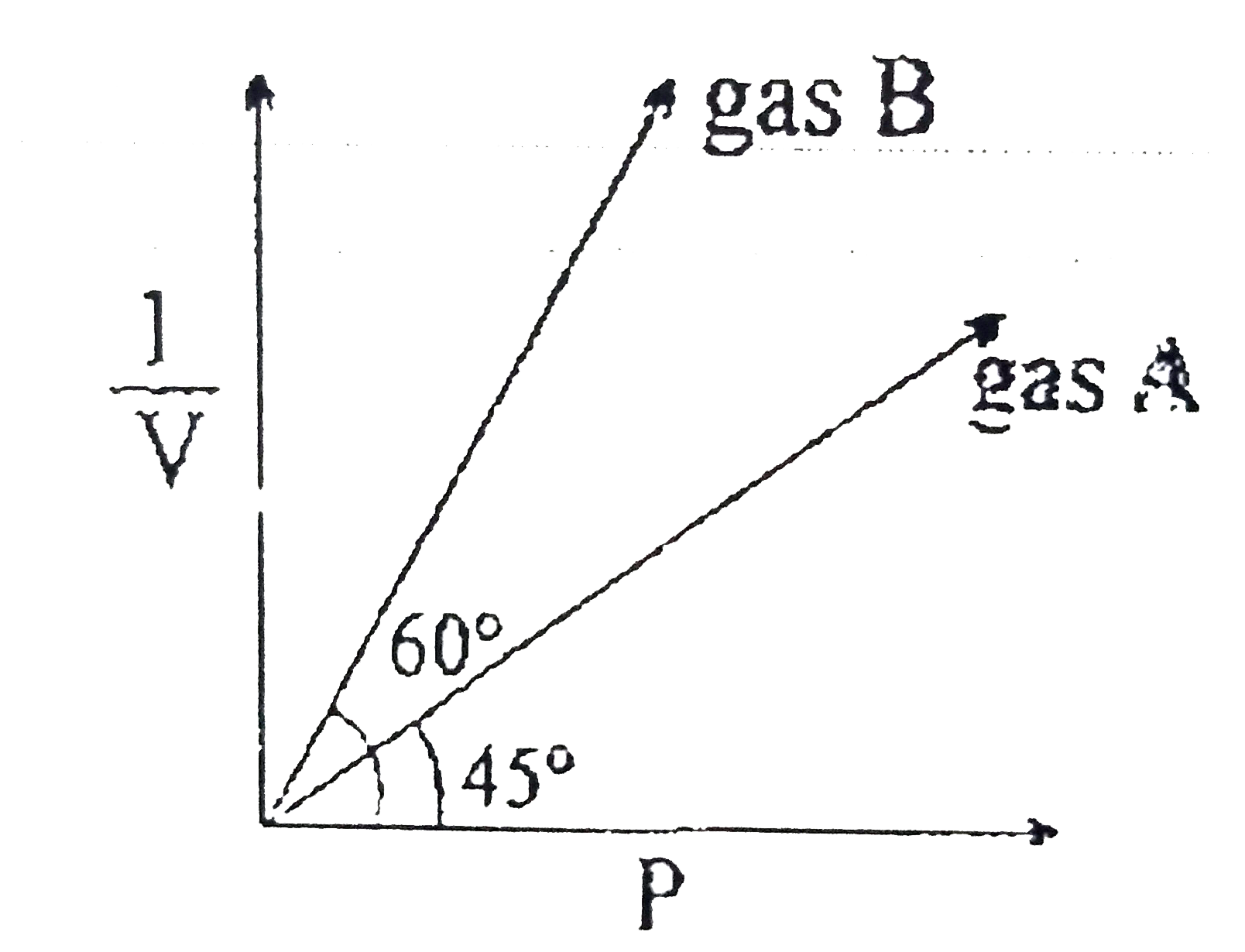
At constant temperature of 273K. (1)/(v) vs are plotted for two ideal

Gaseous State, PDF, Gases

The graph of compressibility factor Z vs P for one mole of a real

The given graph represents the variation of compressibility factor
⏩SOLVED:The behaviour of a real gas is usually depicted by

If the slope of 'Z' (compressibility factor) vs. 'p' curve is constant

Level 3 Passage 3 from NARENDER AVASTHI CHEMISTRY (ENGLISH

What is compressibility factor? What is its value for ideal gas