Mean Free Path, Molecular Collisions
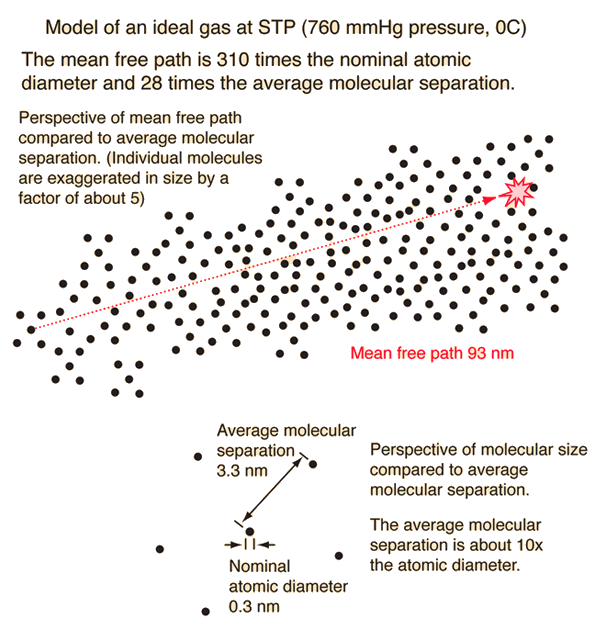

Mean free path and diffusion - ppt download

Mean free path & collision frequency (derivation) - tec-science

What is the relationship between the mean free path and temperature at a constant volume? - Quora

Estimate the mean free path and collision frequency of a nitrogen molecule in a cylinder containing nitrogen at 2.0 atm and temperature 17 °C. Take the radius of a nitrogen molecule to
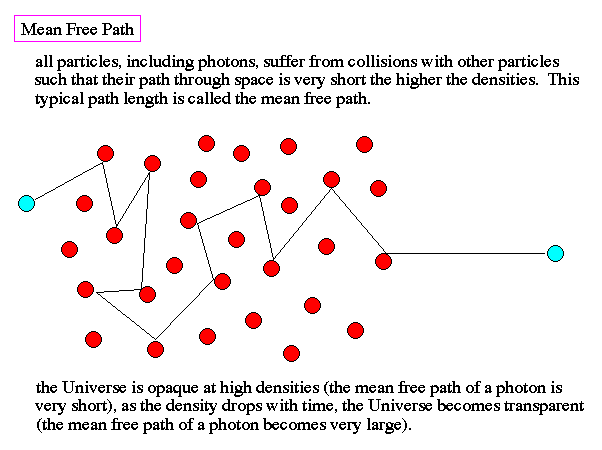
baryon fraction, CMB

Mean Free Path // Molecular Collisions Video 1 // Thermal Physics Semester 3

SOLVED: Calculate the mean free path of a particle in the gas phase of a deposition system and estimate the number of collisions it experiences in traveling from the source to the
20. The mean free path of benzene molecule is6 x 10 6 m and average speed of molecules is1.2 x 103 m/s. The collision rate of molecules, is(1) 3 x 107 s

Why in gases, interatomic distance is of order of tens of A° but mean free path of 1000s of A°? - Quora

Mean Free Path, PDF, Gases

MEAN FREE PATH